Engineering Excellence in
Pharmaceutical & Chemical Industries
From concept to commissioning, we deliver cost-effective and time-bound total engineering solutions for pharmaceutical and chemical projects across the globe.
FDA & WHO Compliant
Comprehensive Engineering Solutions
cGMP Compliant Designs
Complete Project Management
Validation Documentation
Clean Room & HVAC Designs
Procurement

About us
PharmaChem Consultants is an evolution of C&P Engineering Consultants Pvt. Ltd., established in 2000. Born from decades of hands-on experience with leading multinational pharmaceutical and chemical companies, we understand the intricate challenges of modern pharmaceutical engineering.
Founded in 2009, PharmaChem Consultants brings together a dedicated team of engineers, pharmacists, and architects with extensive track records in conceptualizing, designing, and executing complex pharmaceutical and chemical projects.
Our Expertise
Comprehensive Engineering Services
End-to-end solutions for pharmaceutical, chemical, and biotech facilities with expertise spanning four decades
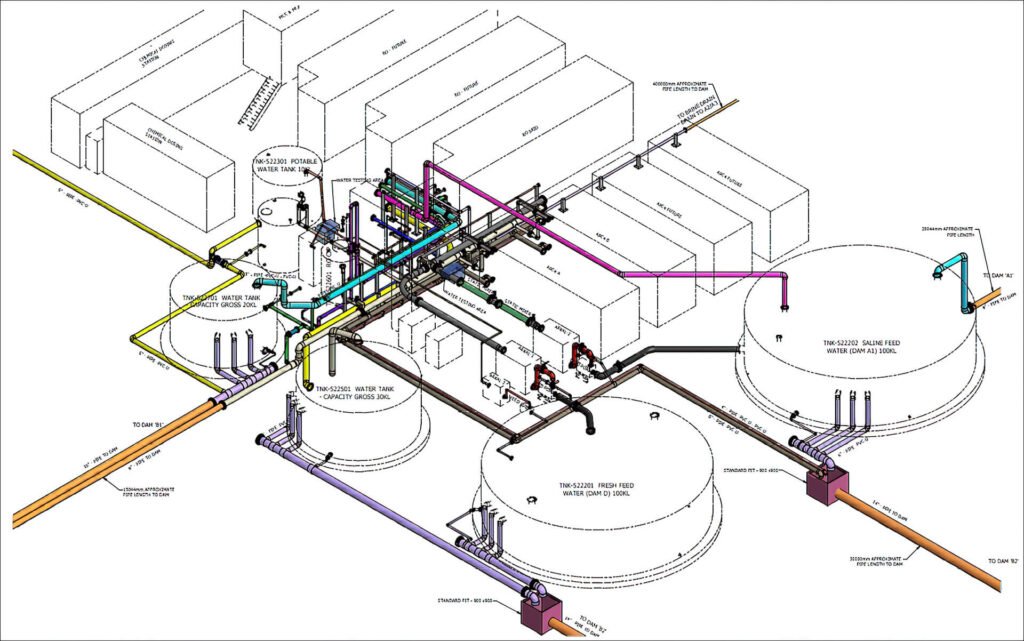
Plant Design & Engineering
Complete conceptualization, basic and detailed engineering for pharmaceutical formulation plants, bulk drug facilities, and chemical processing units.

HVAC & Clean Room Systems
Design and implementation of industrial HVAC systems, clean rooms, aseptic areas, and environmental control systems meeting international standards.

Project Management
Comprehensive project coordination from planning to commissioning, ensuring time and cost efficiency with quality compliance.

Utilities & Infrastructure
Design and setup of purified water systems, WFI, clean steam, utilities, and complete infrastructural facilities for industrial operations. Like warehouses , quality control, laboratories and other supporting facilities

Laboratory Design
Specialized design for R&D, analytical, and quality control laboratories with proper ventilation, utilities, and safety systems.

Validation & Documentation
Preparation of validation documents, as-installed drawings, and compliance documentation for regulatory requirements including FDA, WHO, and cGMP.
Why PharmaChem
What Sets Us Apart
Four decades of engineering excellence combined with deep industry knowledge and commitment to quality
Proven Track Record
Successfully delivered 200+ projects across 15+ countries for leading pharmaceutical and chemical companies.
Expert Team
Conglomeration of experienced engineers, pharmacists, and architects with specialized domain expertise.
Procurement
Preparation of specifically for purchase of plant, machineries and another items & provide assistance for procurement
Global Experience
International project experience including FDA and MHRA approved facilities in multiple continents.
Cost Effective
Optimized designs and efficient project management ensuring maximum value within budget constraints.
Compliance Focused
Deep understanding of cGMP, WHO, FDA, MHRA requirements ensuring regulatory compliance from day one.
Our Clients
Trusted by Industry Leaders
We’ve partnered with leading pharmaceutical and chemical companies worldwide

Serum Institute of India

Charak Pharmaceuticals Pvt. Ltd

Chiron Behring Vaccines Pvt Ltd

Medico Remedies Limited

Sanofi India Limited

Raman And Weil Pvt Ltd

Biladi Pharma Ltd

Prochem Pharmaceuticals Pvt. Ltd.

Bharat Serums and Vaccines Limited
Why PharmaChem
Ready to Start Your Project?
Let’s discuss how we can help you build world-class pharmaceutical or chemical facilities that meet international standards
